 
The uncertainty principle implies that it is in general not possible to predict the value of a quantity with arbitrary certainty, even if all initial conditions are specified. 
Such variable pairs are known as complementary variables or canonically conjugate variables and, depending on interpretation, the uncertainty principle limits to what extent such conjugate properties maintain their approximate meaning, as the mathematical framework of quantum physics does not support the notion of simultaneously well-defined conjugate properties expressed by a single value. The Uncertainty principle is also called the Heisenberg uncertainty principle. 
Origin of Heisenberg Uncertainty Principle Named for Werner Heisenberg, its discoverer, translated into English from a variety of original German terms. This relation expresses Heisenberg’s uncertainty principle. Heisenberg Uncertainty Principle definition: Uncertainty principle. The product of the uncertainty in position of a particle and the uncertainty in its momentum can never be less than one-half of the reduced Planck constant: (4.3.1) x p 2. In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physical quantities of a particle, such as position, x, and momentum, p, can be predicted from initial conditions. According to Heisenberg, these uncertainties obey the following relation. Σ x σ p ≥ ℏ 2 is called the momentum operator in position space.Wikipedia Rate this definition: 0.0 / 0 votes The formal inequality relating the standard deviation of position σ x and the standard deviation of momentum σ p was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928: In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ pΔ q ≈ h using the full Planck constant. Introduced first in 1927 by German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. Such paired-variables are known as complementary variables or canonically conjugate variables. In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the product of the accuracy of certain related pairs of measurements on a quantum system, such as position, x, and momentum, p. In short: the uncertainty principle describes a trade-off between two complementary properties, such as speed and position. Later, in 1927, a German physicist called Werner Heisenberg proposed that it is not possible to know where exactly an electron is located when it behaves like a wave and what its velocity is simultaneously because by trying to take any measurements, we would be disturbing it in some way. The uncertainty principle in its standard form describes how precisely we may measure the position and momentum of a particle at the same time if we increase the precision in measuring one quantity, we are. If the operators A and B are scalar operators (such as the position operators) then AB BA and the commutator is always zero. The uncertainty principle has been frequently confused with the observer effect, evidently even by its originator, Werner Heisenberg. Read on and understand the thought process of this great. What is the position of a particle, such as an electron Is it at the center of the wave The answer lies in how you measure the position of an electron. Probability Distribution Matter and photons are waves, implying they are spread out over some distance. The principle is named after German physicist Werner Heisenberg, who proposed the uncertainty principle in the year 1927. Thus, there is uncertainty in all kinds of measurements because of the property of a particle. Explain the implications of Heisenberg’s uncertainty principle for measurements. Heisenberg’s uncertainty principle states that for particles exhibiting both particle and wave nature, it will not be possible to accurately determine both the position and velocity at the same time. 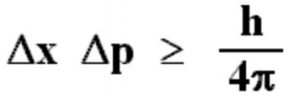
It states that there is a variety of mathematical inequalities in measurements which creates a kind of fundamental limit to the precision. Meaning of heisenberg uncertainty principle. Heisenberg’s Uncertainty Principle: Definition. Uncertainty principle of Heisenberg, 1927. Heisenberg Uncertainty Principle Definition. The Commutator of two operators A, B is the operator C A, B such that C AB BA. Definition of heisenberg uncertainty principle in the dictionary. Canonical commutation rule for position q and momentum p variables of a particle, 1927. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
